
 ABT-263 (Navitoclax)
ABT-263 (Navitoclax)
S1001
5~200mg
Brand
Selleckchem
Description
ABT-263 (Navitoclax)
ABT-263 (Navitoclax) is a potent inhibitor of Bcl-xL, Bcl-2 and Bcl-w with Ki of ≤ 0.5 nM, ≤1 nM and ≤1 nM, but binds more weakly to Mcl-1 and A1. Phase 1/2.
 Distributor Tel: +886-2-2827-1197jimmy@new.abscience.com.tw
Distributor Tel: +886-2-2827-1197jimmy@new.abscience.com.tw
Biological Activity
| Description | ABT-263 (Navitoclax) is a potent inhibitor of Bcl-xL, Bcl-2 and Bcl-w with Ki of ≤ 0.5 nM, ≤1 nM and ≤1 nM, but binds more weakly to Mcl-1 and A1. Phase 1/2. | |||||
|---|---|---|---|---|---|---|
| Targets |
Bcl-xL |
Bcl-2 |
Bcl-w |
|||
| IC50 |
≤ 0.5 nM (Ki) |
≤1 nM (Ki) |
≤ 1 nM (Ki)[1] |
|||
| In vitro | ABT-263 is structurally related to ABT-737; it is a disruptor of Bcl-2/Bcl-xL interactions with pro-apoptotic proteins. Overexpression of the prosurvival Bcl-2 family members is commonly associated with tumor maintenance, progression, and chemoresistance. [1] ABT-263 displays the protection afforded by overexpression of Bcl-2 or Bcl-xL with EC50 values of 60 nM and 20 nM, respectively. [1] A wide range of cellular activity is observed with ABT-263 having a 50% growth inhibition (EC50) of 110 nM against the most sensitive line (H146), whereas its activity in the least sensitive line (H82) results in an EC50 at 22 μM. All four cell lines with EC50 values of <400 nM (H146, H889, H1963, and H1417) are also highly sensitive to ABT-737, and the two most resistant lines (H1048 and H82) are similarly resistant to ABT-263. [2] | |||||
| In vivo | When ABT-263 is administered at 100 mg/kg/day in the H345 xenograft model, significant antitumor efficacy is observed with 80% TGI and 20% of treated tumors indicating at least a 50% reduction in tumor volume. [2] Oral administration of ABT-263 alone causes complete tumor regressions in xenograft models of small-cell lung cancer and acute lymphoblastic leukemia. In xenograft models of aggressive B-cell lymphoma and multiple myeloma where ABT-263 displays modest or no single agent activity, it significantly enhances the efficacy of clinically relevant therapeutic regimens. [2] | |||||
| Features | ||||||
Protocol(Only for Reference)
Kinase Assay:
| Affinity determination | Binding affinities (Ki or IC50) of ABT-263 against different isoforms of Bcl-2 family are determined with competitive fluorescence polarization assays. The following peptide probe/protein pairs are used: f-bad (1 nM) and Bcl-xL (6 nM), f-Bax (1 nM) and Bcl-2 (10 nM), f-Bax (1 nM) and Bcl-w (40 nM), f-Noxa (2 nM) and Mcl-1 (40 nM), and f-Bax (1 nM) and Bcl-2-A1 (15 nM). Binding affinities for Bcl-xL are also determined using a time-resolved fluorescence resonance energy transfer assay. Bcl-xL (1 nM, His tagged) is mixed with 200 nM f-Bak, 1 nM Tb-labeled anti-His antibody, and ABT-263 at room temperature for 30 min. Fluorescence is measured on an Envision plate reader using a 340/35 nm excitation filter and 520/525 (f-Bak) and 495/510 nm (Tb-labeled anti-His antibody) emission filters. |
|---|
Cell Assay:
| Cell lines | SCLC cell lines |
|---|---|
| Concentrations | 0-1 μM |
| Incubation Time | 48 hours |
| Method |
Human tumor cell lines SCLC cell lines are maintained at 37 °C containing 5% CO2. SCLC cell lines are cultured in RPMI 1640 with 10% fetal bovine serum (FBS), 1% sodium pyruvate, 25 mM HEPES, 4.5 g/L glucose, and 1% penicillin/streptomycin. Leukemia and lymphoma cell lines are cultured in RPMI 1640 supplemented with 10% FBS and 1% penicillin/streptomycin. Cells (1-5×10 4) are treated by ABT-263 for 48 hours in 96-well culture plates in a final volume of 100 μL and cytotoxicity is assessed with the CellTiter Glo assay. In vitro cyto toxicity of ABT-263 is assayed. |
Animal Study:
| Animal Models | C.B.-17 scid-bg or C.B.-17 scid mice |
|---|---|
| Dosages | 100 mg/kd/d |
| Administration | Administered via p.o. |
References
Clinical Trial Information( data from http://clinicaltrials.gov)
| NCT Number | Recruitment | Conditions | Sponsor /Collaborators |
Start Date | Phases |
|---|---|---|---|---|---|
| NCT01121133 | Completed | Lymphoma, Including Chronic Lymphocytic Leukemia|Solid Tumors | Abbott | 2010-05 | Phase 1 |
| NCT01087151 | Completed | Chronic Lymphocytic Leukemia | Genentech|AbbVie (prior sponsor, Abbott) | 2010-08 | Phase 2 |
| NCT01423539 | Withdrawn | Lymphoma, B-Cell | Genentech|AbbVie (prior sponsor, Abbott) | 2011-10 | Phase 2 |
| NCT01557777 | Active, not recruiting | Chronic Lymphocytic Leukemia | AbbVie|AbbVie (prior sponsor, Abbott) | 2012-06 | Phase 2 |
| NCT01828476 | Not yet recruiting | Castrate Refractory Prostate Cancer (CRPC) | University of Medicine and Dentistry New Jersey|AbbVie | 2013-04 | Phase 2 |
Chemical Information
Download ABT-263 (Navitoclax) SDF
| Molecular Weight (MW) | 974.61 |
|---|---|
| Formula |
C47H55ClF3N5O6S3 |
| CAS No. | 923564-51-6 |
| Storage | 3 years -20℃Powder |
|---|---|
| 6 months-80℃in DMSO | |
| Syonnyms | N/A |
| Solubility (25°C) * | In vitro | DMSO | 100 mg/mL (102 mM) |
|---|---|---|---|
| Water | <1 mg/mL (<1 mM) | ||
| Ethanol | <1 mg/mL (<1 mM) | ||
| * <1 mg/ml means slightly soluble or insoluble. * Please note that Selleck tests the solubility of all compounds in-house, and the actual solubility may differ slightly from published values. This is normal and is due to slight batch-to-batch variations. |
|||
| Chemical Name |
|---|
Preparing Stock Solutions
| Stock Solution (1ml DMSO) | 1mM | 10mM | 20mM | 30mM |
|---|---|---|---|---|
| Mass(mg) | 0.97461 | 9.7461 | 19.4922 | 29.2383 |
Customer Reviews (12)
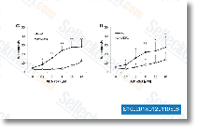 Click to enlarge |
Rating |
|
| Source | Biochem Biophys Res Commun, 2011, 408(2), 344-9. ABT-263 (Navitoclax) purchased from Selleck | |
| Method | Flow cytometry | |
| Cell Lines | MDCKII wild type cells/MDR1 cells | |
| Concentrations | 50 nM | |
| Incubation Time | 24 h | |
| Results | MDCKII cells transfected with MDR1 showed significantly less apoptosis inducedby ABT-737 (Fig. C) and ABT-263 (Fig. D) than wild type cells. |
Product Citations (20)
-
Targeting SOD1 reduces experimental non-small-cell lung cancer. [Glasauer A, et al. J Clin Invest 2013;10.1172/JCI71714]
-
BCL2/BCL-XL inhibition induces apoptosis, disrupts cellular calcium homeostasis, and prevents platelet activation. [Vogler M, et al. Blood 2011;117(26):7145-54]
-
Both TMEM16F-dependent and TMEM16F-independent pathways contribute to phosphatidylserine exposure in platelet apoptosis and platelet activation. [van Kruchten R, et al. Blood 2013;121(10):1850-7]
Application
Reactivity



